 
This was the beginning of further understanding leading to the atomic theory and structure that we know today.Ĭathode ray tube is a tube that contains a small amount of gasīetween two metallic plates.Click on a date/time to view the file as it appeared at that time. Thomson found that cathode rays always had the same e/m ratio, no matter what metals were used for the cathodes and no matter what gas was used in the tubes. 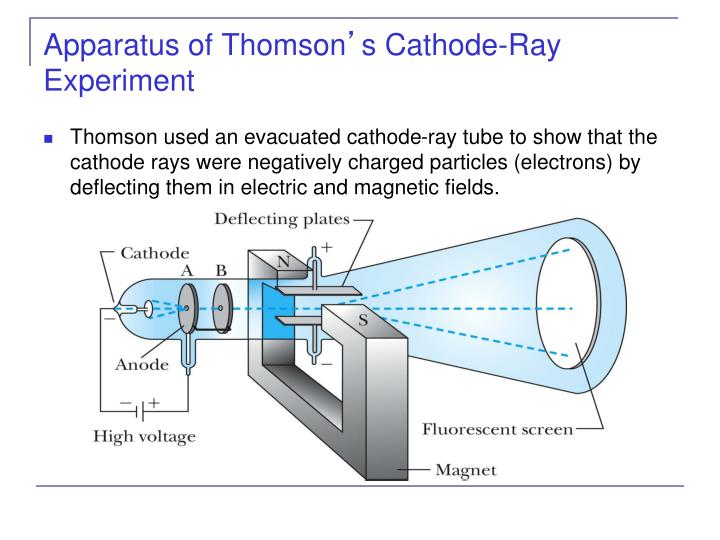 
He won the 1906 Nobel prize for the discovery of electrons. The gold foil experiment is one of the most. Cathode rays are deflected by both magnetic and electric fields. Three big experiments that helped shape the electron and the atom concept that we have today are the discovery of the electron and the plum pudding model, Rutherford's gold foil experiment, and the Millikan oil-drop experiment. The cathode ray discharge tube experiment does not lead to the discovery of radioactivity. Thomson measured the deflection of the beam using a ruler etched on the end of. You can see a simulation of this glow on the far right of the applet diagram, as shown in Figure 2. de 2020 As he flowed the current, he saw that some invisible rays move from the. In 1897 JJ Thomson discovered the electron by using cathode-ray tubes. In Thomson’s experiment, a fluorescent material was coated on the end of the tube to produce a glowing dot where the cathode rays hit. He zapped atoms with electricity and observed that negatively charged particles were removed! He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. In 1897, Thomson showed that cathode rays were composed of previously unknown negatively charged particles (now called electrons), which he calculated must. Cathode Ray Tube Experiment by J.J Thomson full explained Web25 de oct. The results of these experiments helped Thomson determine the mass-to-charge ratio of the cathode ray particles, which led to a fascinating discovery - minus the mass of each particle was much, much smaller than that of any known atom. Thomson, a British physicist, conducted the cathode ray experiment. Thomson also placed two magnets on either side of the tube, and observed that this magnetic field also deflected the cathode ray. Millikan discovered that there is a fundamental electric chargethe charge of an electron. Thomson’s cathode ray tube showed that atoms contain small, negatively charged particles called electrons. Millikan discovered that there is a fundamental. Thomson used a cathode ray tube to determine the nature of the. Thomsons cathode ray tube showed that atoms contain small, negatively charged particles called electrons. Experiments showed that the rays had mass. Thomsons cathode ray tube experiment-an experiment in which the electron was discovered, along with its charge-to-mass ratio. The Thompson Cathode Ray Experiment was an experiment conducted by J. Previously, atoms were known to be indivisible, but in 1897, J. The cathode ray tube was first invented by Sir William Crookes. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
